Real-world data (RWD) is not a new idea in the healthcare industry, but what is new is how it continues to evolve and the untapped potential it holds. Still, what is RWD, and what can it reveal about patient journeys? According to the Food and Drug Administration (FDA), “RWD is the data relating to patient health status or the delivery of health care routinely collected from a variety of sources such as, electronic health records, claims and billing activities, registries, etc.” Real-world data is information, typically, generated outside of or after a clinical trial. It can include claims or clinical data, as well as any data that augments a patient’s medical experiences and better reflects what is really happening in their real world – not just a more controlled setting.
It is important to understand and define the closely related, term real-world evidence (RWE) as well. As per the FDA, RWE is the clinical evidence regarding the usage and potential benefits or risks of a medical product derived from RWD. You cannot have RWE without RWD and not all RWD turns into evidence.
As more data assets become available and the regulatory use of real-world data and evidence continues to grow and expand, the true impact that this data truly holds is changing. Life sciences, pharmaceutical, and medical device companies can all use real-world data to their benefit.
In a webinar sponsored by LexisNexis® Risk Solutions, and hosted by Informa, we expand on the evolution of RWD, as well as the impact that RWD — and tokenization — can have in capturing a more holistic and precise patient journey.
RWD can be critical in revealing valuable connections.
What Types of RWD Exist?
RWD increasingly includes data assets at the individual level. These assets are not limited to the medical or clinical experience. Rather, they might include a person’s wearable data, consumer habits, and other types of information that might affect a person’s experiences with healthcare.
As more data becomes available, it will continue to augment what the industry considers RWD. For example, a few years ago, including images seemed out of reach. Now, images may be included in patient datasets.
Social Determinants of Health
Social determinants of health (SDoH) stand to add even greater value. Did you know that social factors account for more than 33% of total deaths annually in the U.S.? There is enormous untapped potential in SDoH that the industry is only beginning to realize, including:
- Healthcare access and quality
- Neighborhood and built environment
- Social and community context
- Economic stability
- Education and access quality
Mortality Data
Mortality events may go unrecognized in a clinical study. The FDA states, in their draft guidance on real-world evidence, “excluding patients who appear to be lost to follow-up at any time following their exposure to the study drug is likely to create bias.” In fact, they estimate that 35% of deaths are missing from EHR data. Some solutions currently in use, such as the National Death Index and Social Security Index, face challenges including significant lag time. A comprehensive measure of mortality data is crucial.
Thinking about life events as a normal part of analysis and research, instead of the exception, can help life sciences organizations gain more detailed, meaningful views into people and their lives and understand how these nuances might impact research results.
Data Tokenization Enables De-Identified Use Cases
Tokenization enables us to view sensitive data elements without revealing the identity of the individual associated with the data. Tokenized data opens new possibilities for analysis and data sharing — all while remaining secure and compliant.
Not all Data Tokenization Options are Equal
Many tokenization options exist in the industry today. These are based on observed input PII data to create a unique, de-identified patient token. However, these techniques are only as good as the information received. Incorrectly linking individuals with some matching information, or missing matches due to changes in a person’s situation can lead to inaccuracies. The FDA has weighed in on data linking. In their guidance, they discuss probabilistic and deterministic approaches to data linkage, which can be applied differently, depending on the use case. Probabilistic and deterministic matching both incur risk in assembling an incomplete view of individuals.
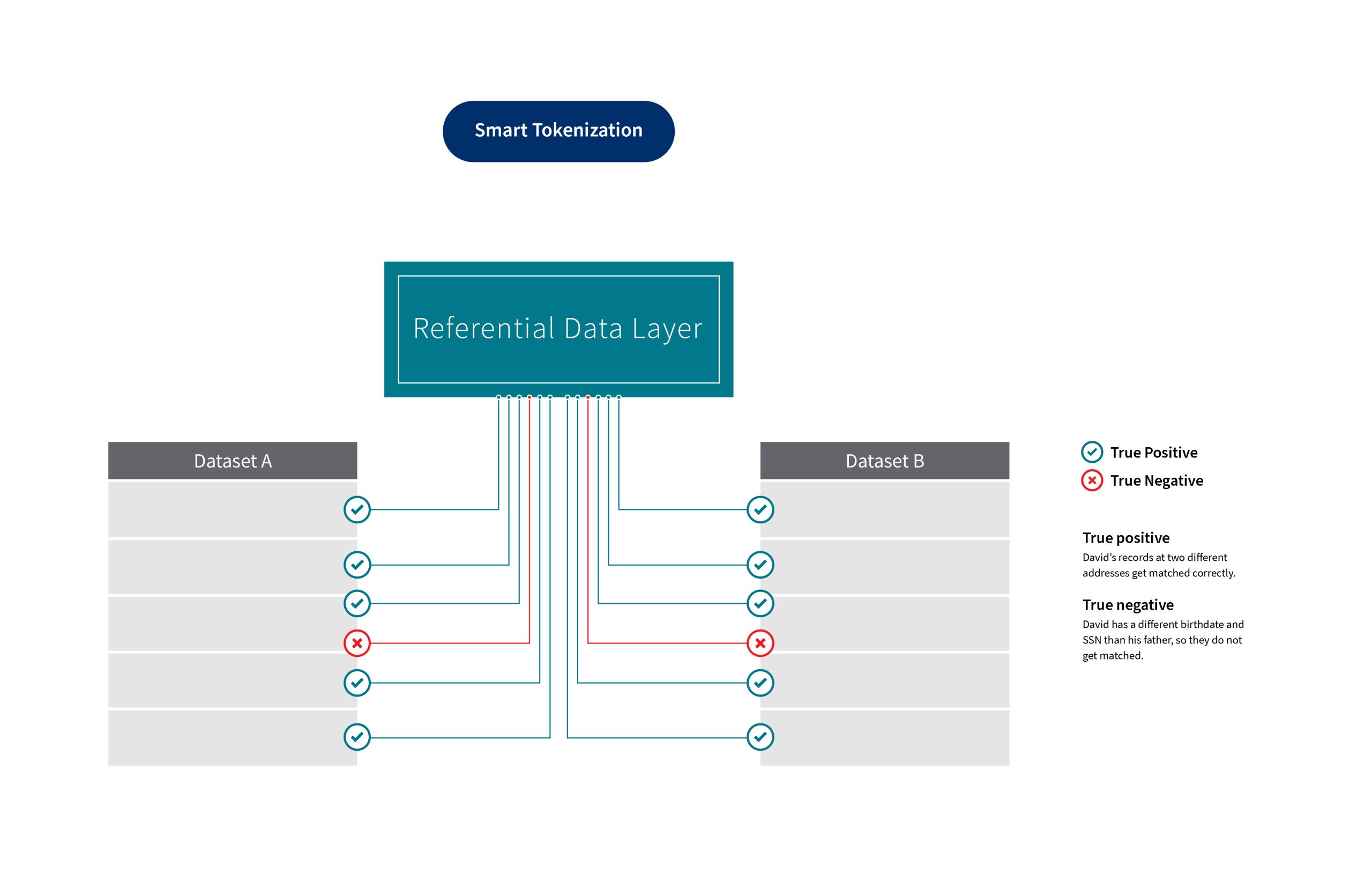 The LexisNexis®Gravitas™ solution adds a Referential Data Layer to create a next-generation data tokenization solution. This solution allows for greater confidence, consistency, and precision in data matching. It works across different types of datasets and over time. People’s lives change in real time — moving, getting married, changing jobs, and more — so the work of matching and de-identifying data is never ending.
The LexisNexis®Gravitas™ solution adds a Referential Data Layer to create a next-generation data tokenization solution. This solution allows for greater confidence, consistency, and precision in data matching. It works across different types of datasets and over time. People’s lives change in real time — moving, getting married, changing jobs, and more — so the work of matching and de-identifying data is never ending.
Real-World Integration of Healthcare Tokenization
Information coming in from numerous, disparate data sources plays a crucial role in creating a more precise and a more complete patient view. Social determinants of health and mortality data feed into that complete view as well. What information we consider and how we link it together directly impacts results – and in healthcare, precision matters. Anything that is unmeasured or imperfectly measured will limit the impact data can have on improving patients’ lives. A de-identified solution lets us examine the whole person’s experience.
These results can drive communications strategy, pricing, decision support tools, clinical trial design, and much more.





